When FDA inspectors walk into a juice production space, they are not just checking surfaces for cleanliness. They are reading how work moves. Where raw produce enters, where hands change tasks, where waste exits, and whether finished juice is protected from earlier risks. This is why an FDA compliant workflow matters as much as ingredients, equipment, or documentation.
For many juice businesses in Ghana, especially those scaling from home kitchens into commercial spaces, the U-shaped workflow offers a practical way to control risk without requiring a large or expensive facility. It works not because it is fashionable, but because it aligns naturally with how regulators think about contamination, process control, and operational discipline.
This post breaks down how to design and run a U-shaped workflow that holds up under real inspection conditions in Ghana.
Why Workflow Design Matters More Than Floor Size
One of the most common misconceptions about compliance is that space solves problems. In reality, inspectors are more interested in behaviour than square metres.
A small room with a disciplined workflow often performs better during inspection than a larger space where movement is chaotic. When staff repeatedly cross from dirty to clean zones, when raw produce passes through bottling areas, or when waste exits through production paths, risk increases. Even if nothing goes wrong that day, the potential is visible.
This is why meeting FDA expectations as a growing juice business often starts with layout thinking, not expansion. Workflow quietly communicates intent. It tells regulators whether hygiene is enforced by habit or by chance.
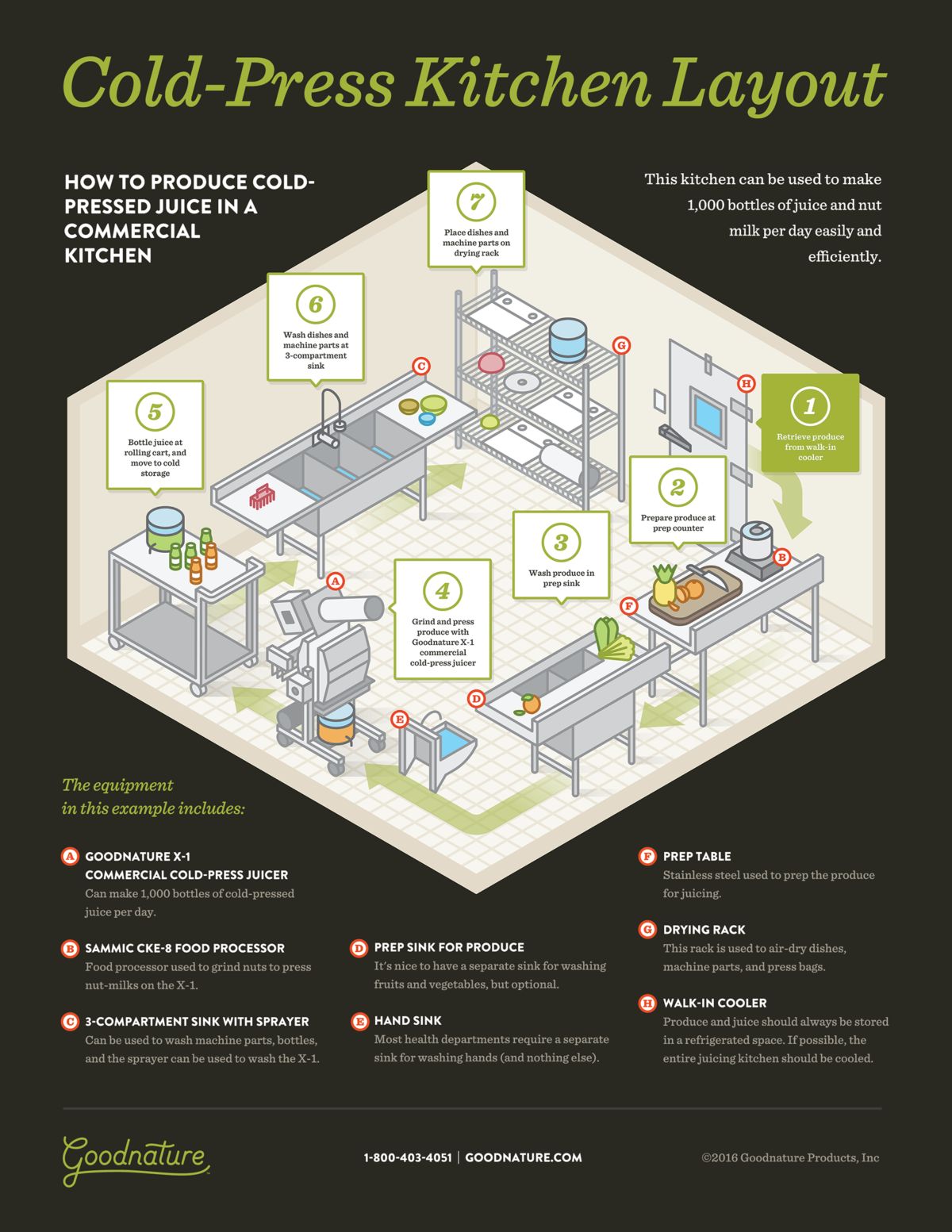
Understanding the Logic Behind a U-Shaped Workflow
A U-shaped workflow is built around controlled progression. Production begins at one end of the “U” with the highest contamination risk and gradually moves toward the lowest-risk stage without reversal.
Raw fruits enter, are washed, processed, bottled, and then moved into cold storage or dispatch. At no point should finished juice need to travel back past earlier stages. People move forward with the product, not against it.
This mirrors applying GMP principles in real juice production environments, where segregation, directional flow, and reduction of cross-contact are core ideas. The shape itself is not magic. The discipline it enforces is.
Applying a U-Shaped Workflow in Typical Ghanaian Spaces
Most juicepreneurs in Ghana are not working with purpose-built factories. They operate from shops, containers, converted rooms, or compact commercial kitchens.
This is where the U-shaped workflow becomes especially useful. Instead of spreading activities across the room, the process wraps along the walls. One side becomes intake and washing. The curve handles processing and bottling. The final arm leads directly to refrigeration or dispatch.
This approach works with existing water points, drainage, and access limitations rather than fighting them. It also forces clarity around where each activity belongs, which reduces improvisation over time.
However, workflow design at this stage must also account for assessing utility load before scaling production. Placing high-load equipment like cold-press machines or chest freezers too close together, or late in the flow without electrical planning, can overload circuits and introduce safety risks that undermine both compliance and continuity.
How the Workflow Supports FDA Expectations
FDA inspectors are trained to assess risk patterns. When workflow is unclear, inspections become interrogations. When flow is logical, inspections become confirmations.
An FDA compliant workflow reduces the likelihood of raw and finished products crossing paths. It supports traceability because each stage is clearly defined. It also makes hygiene lapses easier to spot and correct internally before they attract regulatory attention.
This is where documenting SOPs for consistent juice production stops being paperwork and starts becoming visible practice. When SOPs describe a process that the space naturally enforces, compliance becomes easier to sustain.
Workflow as a Physical Extension of SOPs and GMPs
SOPs and GMPs describe what should happen. Workflow determines what actually happens.
If your SOP says fruits must be washed before processing, but the processing table is easier to reach than the wash station, staff will follow convenience. A U-shaped layout removes that conflict. It makes the correct action the easiest action.
This alignment between documentation and physical design is one of the quiet indicators regulators use to assess operational maturity. When space and procedure reinforce each other, inspectors see control rather than supervision.
Staff Behaviour Improves When Movement Is Clear
People follow paths of least resistance. When workflow is unclear, shortcuts appear. When movement is intuitive, discipline improves without constant reminders.
In Ghanaian production environments where staff turnover or role rotation is common, layout becomes a silent trainer. New staff understand where they should and should not be simply by how the space behaves.
This is why building staffing systems that scale with your business often starts with physical design. Clear workflow reduces training time, limits mistakes, and protects consistency even when supervision is light.
Hidden Risks: Insurance and Liability Exposure
Poor workflow does not only attract FDA questions. It also increases liability.
Cross-contamination incidents, slips, equipment damage, and product recalls often trace back to early layout decisions. This is where workflow quietly supports how insurance protects a growing juice business. Insurers assess operational risk in ways that closely mirror regulatory logic.
A disciplined layout lowers exposure long before a claim is ever filed.
When to Revisit the Workflow as You Grow
As production volume increases, workflow stress usually appears before financial stress. Bottlenecks form, staff improvise, and contamination risk creeps back in.
Revisiting your U-shaped layout at each growth stage helps maintain alignment with SOPs, GMPs, FDA expectations, staffing realities, insurance exposure, and power capacity. This is especially important when transitioning from a home kitchen into a commercial space, where informal habits quickly become visible weaknesses.
Workflow should evolve deliberately, not reactively.
Designing flow with clarity and confidence
An FDA-compliant U-shaped workflow is not about aesthetics or copying industrial factories. It is about intentional movement, controlled risk, and making it easy to do the right thing every day. When workflow is designed properly, hygiene improves naturally, staff make fewer mistakes, and inspections feel less confrontational. When ignored, even clean spaces raise uncomfortable questions.
The goal is not to impress regulators. It is to protect your product and your process.
Join the Juicepreneurs Community
Inside the Juicepreneurs Community, juicepreneurs share real experiences about workflow design, layout corrections, FDA inspection feedback, and the small changes that made a big difference in compliance. You learn how others adapted U-shaped workflows to tight Ghanaian spaces and what inspectors actually focused on during visits. This shared insight helps you avoid costly trial-and-error and build with confidence.
Book a One-on-One Consultation
If you want help assessing your current production layout, correcting workflow weaknesses, or designing an FDA-compliant U-shaped workflow that fits your exact space and production volume, a one-on-one consultation allows us to walk through it together. The focus is practical adjustments that align with SOPs, GMP expectations, staffing behaviour, power readiness, and real inspection standards in Ghana.
Download the Juicepreneur Blueprint
The Juicepreneur Blueprint brings structure to workflow design, SOPs, GMP discipline, FDA compliance thinking, facility readiness, staffing systems, insurance, and growth planning. It is designed to help juice businesses build production systems that regulators trust and teams can actually follow, without overcomplicating operations.
Good workflow does not announce itself. It quietly keeps everything in order.
For new juicepreneurs, I have put together what I call the must-read list of posts on this site to get you started on your business journey:
- Read about juicing equipment here.
- Read about the different types of pineapples here.
- Get beginner insight into beverage catering here.
- Read about record keeping in the juice business here.
- If you have already started beverage catering, read about costly mistakes to avoid here.
- Learn where to source PET bottles and other essentials here.
- Learn how to write a juice business plan here and here.
- Training new staff can be a headache, learn how to build a system to help you here.
- The Norwalk Juicer is a very fine machine, its not for everyone though. Learn more here.
- The juice business is heavily dependant on suppliers. Learn how to build a relaible network of supplier here.
- FDA compliance is a key metric in this business. Learn how to register your juice products with the FDA here
- Employing Staff can’t be avoided as you grow your business, learn how to build a staffing system that meets your needs and grows with your business here.
- Logistic is very vital in the juice busines, learn about it here.
- Learn how to start a juice truck business here.
- If you are just starting out and need a good but affordable slow juicer for your business. Check out the German Chef Slow Juicer.
- The food handler certification is a must for all your staff including yourself, learn how to secure them here.
- Lastly, read about how to price your beverage catering business here.

